

Mobile/Wechat/WhatsApp: 18592031892
Email:hiwangqiang@163.com
InChI:InChI=1/C28H37N5O7/c1-17(2)12-23(28(39)40)33-27(38)22(14-18-6-4-3-5-7-18)32-25(36)16-30-24(35)15-31-26(37)21(29)13-19-8-10-20(34)11-9-19/h3-11,17,21-23,34H,12-16,29H2,1-2H3,(H,30,35)(H,31,37)(H,32,36)(H,33,38)(H,39,40)/t21-,22-,23-/m0/s1
-
Purpose. To evaluate the cellular permea...
We have designed a new photolabile linke...
An approach using water as a solvent (co...
New fluorous supports were synthesized a...
Seeking a more environmentally balanced ...
The growing interest in synthetic peptid...
The invention relates to a TSBP auxiliar...
A new phosphonium based coupling reagent...
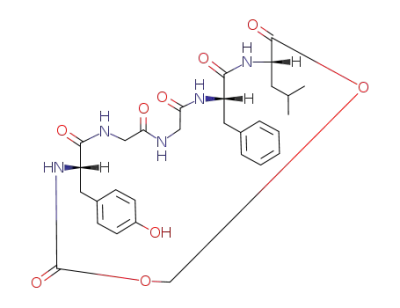
(6S,15S,18S)-15-Benzyl-6-(4-hydroxy-benzyl)-18-isobutyl-1,3-dioxa-5,8,11,14,17-pentaaza-cyclononadecane-4,7,10,13,16,19-hexaone


formaldehyd

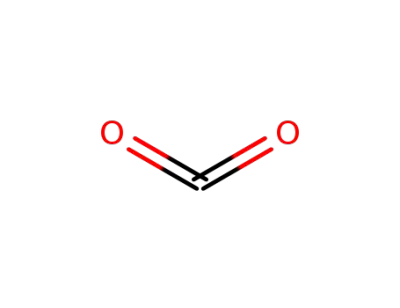
carbon dioxide


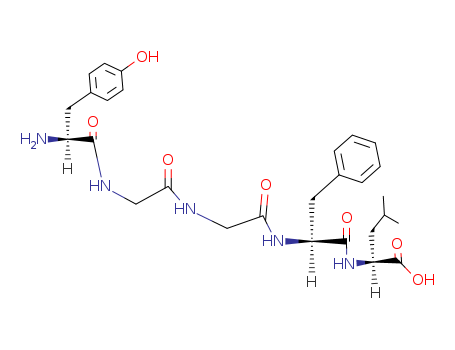
Leu-enkephalin
| Conditions | Yield |
|---|---|
|
rat liver homogenate;
In
water; dimethyl sulfoxide;
at 37 ℃;
pH=7.4;
Further Variations:;
Catalysts;
with esterase inhibitor;
Enzyme kinetics;
|
N-tert-butoxycarbonyl-L-leucine


N-(fluoren-9-ylmethoxycarbonyl)glycine

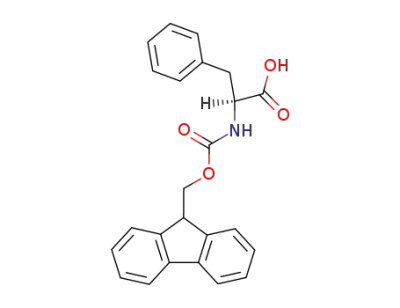
N-Fmoc L-Phe

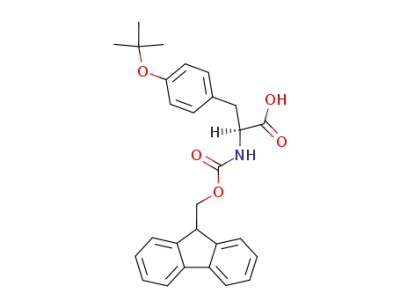
Fmoc-Tyr(tBu)-OH


Leu-enkephalin
| Conditions | Yield |
|---|---|
|
N-tert-butoxycarbonyl-L-leucine;
at 50 ℃;
Ionic liquid;
Inert atmosphere;
With
water; sodium hydroxide;
In
tetrahydrofuran;
N-(fluoren-9-ylmethoxycarbonyl)glycine; N-Fmoc L-Phe; Fmoc-Tyr(tBu)-OH;
Temperature;
Further stages;
|
95% |

O-benzyl-N-tert-butoxycarbonyl-L-tyrosine
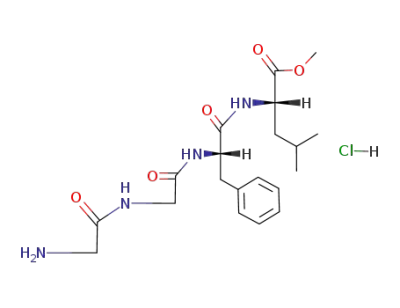
Glycyl-glycyl-(S)-phenylalanyl-(S)-leucinmethylesterhydrochlorid
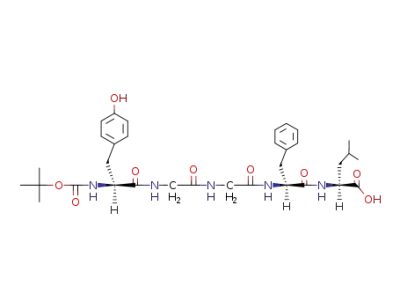
N-tert-butyloxycarbonyl-L-tyrosyl-glycyl-glycyl-L-phenylalanyl-L-leucine
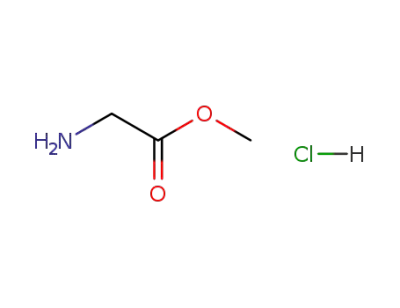
glycine ethyl ester hydrochloride

Q-Tyr-Gly-Gly-Phe-Leu-OH
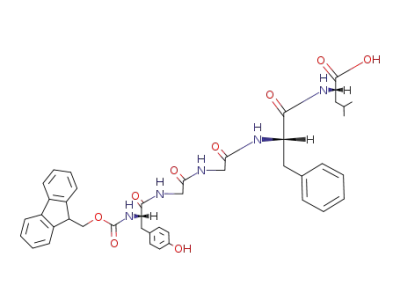
FMOC-L-Tyr-Gly-Gly-L-Phe-L-Leu-OH
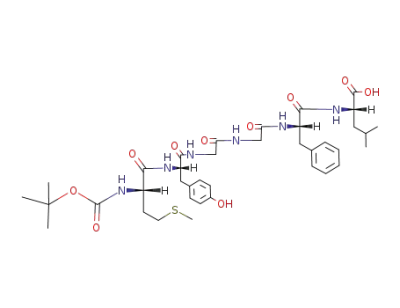
Tert-butyloxycarbonylmethionyl-tyrosyl-glycyl-glycyl-phenylalanyl-leucine
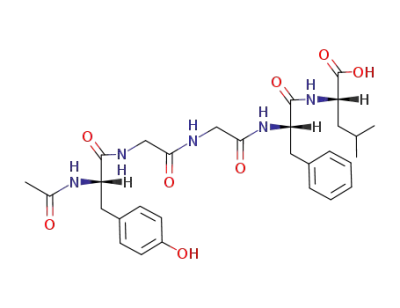
acetyl leucine enkephalin